
GluCloZ's
Pre-Clinical Journey!
The Discovery process of Rapha GluCloZ was inspired by the selection of SouthEast Asian Indigenous herbs and plant foods that are recognized as “GRAS - Generally Recognized as Safe”, an acronym used by the United States Food and Drug Administration (FDA). It refers to a designation that a food substance, chemical or substance added to food is considered safe by experts under the conditions of its intended use.
Thus by definition, GluCloZ qualifies as a non-drug, tea mixture. It can be used freely under the conditions of intended use i.e. for recreation, enjoyment, and as a general benefit to health. Neither does it qualify nor require “Clinical Trials” as the even FDA as with other countries, is not authorized to regulate food.
Teas, including herbal teas, fall under the category of food products. According to the US Federal Food, Drug and Cosmetic Act, importers of food products intended for introduction into US interstate commerce are responsible for ensuring that the products are safe, sanitary, and labeled according to US requirements.
The FDA is not authorized under the law to approve, certify, license, or otherwise sanction individual food importers, products, labels, or shipments. Importers can import foods into the United States without prior sanction by FDA, as long as the facilities that produce, store, or otherwise handle the products are registered with FDA and prior notice of incoming shipments is provided to FDA.
Rapha GluCloZ components


GluCloZ is shown to lower blood glucose response & the glycemic index of foods
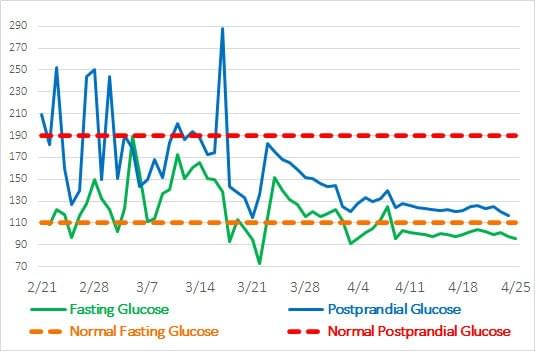
Figure 1. The fasting and postprandial glucose of a diabetic volunteer in 2 months of using GluCloZ
In university tests conducted by Rapha, a single 4 g dose of GluCloZ was incorporated directly into common dietary carbohydrates and given to diabetic individuals. GlucoLimit caused a significant reduction in the glycemic index of the test carbohydrates. The greatest effect was seen with postprandial blood glucose(PBG) tests, where GluCloZ lowered the PBG by 55%. These findings were replicated with continual trials. This reduction in glycemic index of foods results in significant lowering in postprandial blood glucose levels.
A study by Liao et al. (2019), showed the anti-diabetic properties that are in the polysaccharide isolated from abelmoschus esculentus (AE). The polysaccharides were extracted from the AE and were tested on 64 diabetically induced rats on a high fat diet for eight weeks. From this data, we can see that the rats which consumed the AE resulted in a dramatically lower blood glucose level at the end of the test (Figure 2A). Additionally, the AE group had a decrease in food and water consumption (Figure 2C, D) which, resulted in greater weight loss at the end of the tests (Figure 2B).
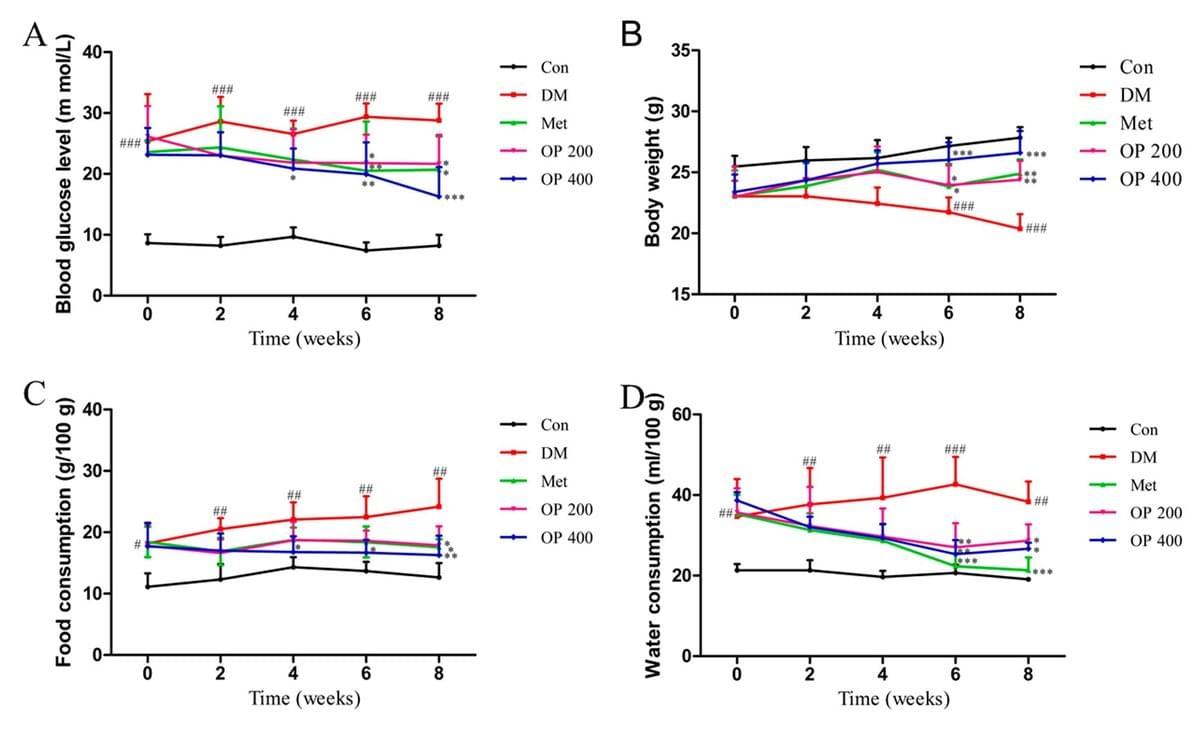
Figure 2. Effects of okra (OP) on fasting blood glucose level (A), body weight (B), food consumption (C) and water consumption (D). Data are expressed as means ± standard deviation (SD; n = 10). Con—control group treated with 0.5% sodium carboxyl methyl cellulose (CMC-Na) per day; DM—type 2 diabetes mellitus (T2DM) treated with 0.5%CMC-Naper day; Met—T2DMmice treated with 200mg/kg metformin per day; OP 200—T2DM mice treated with 200 mg/kg OP per day; OP 400—T2DM mice treated with 400 mg/kg OP per day (Liao et al., 2019)
References.
Liao, Z., J. Zhang, B. Liu, T. Yan, F. Xu, F. Xiao, B. Wu, K. Bi, & Y. Jia (2019). Polysaccharide from Okra (Abelmoschus esculentus (L.) Moench) Improves Antioxidant Capacity via Type 2 Diabetes Model. Molecules, 24(10): 1906.
*These statements have not been evaluated by the US Food and Drug Administration.
This product is not intended to diagnose, treat, cure, or prevent any disease.





